Summary
The modern day labs are pressured on all sides. Deadlines shrink. Rules of compliance become more strict. The clients want quicker reporting. But there are still a lot of teams that use spreadsheets and paper records. That creates workflow bottlenecks, transcription errors, and fragmented data. That gap is bridged by a laboratory information management system. Labs instead of pursuing paperwork consolidate operations and automate the sample tracking. This leads to an increase in productivity and a decrease in compliance risk. Briefly, a LIMS software will transform the chaos of manual work into streamlined organizational workflow in both INDIA and the USA.
Introduction
Firstly, the majority of labs have problems with hidden inefficiencies. Staff copy data manually. Supervisors balance incomparable spreadsheets. Managers approach audits with trepidation. Despite the hard work of teams, there are systems that restrain them.
The paper processes give silos. The use of manual transcription weakens data integrity. Lack of integration slows down turnaround. Consequently, time and confidence are lost by the labs.
Simultaneously, the industry is fast transitioning into automation. According to recent industry market research, the global LIMS software market was valued at USD 2.44 billion in 2024 and is projected to reach USD 3.56 billion by 2030, growing at a CAGR of 6.22%.
These currently lead adoption, while cloud-based LIMS solutions represent the largest deployment segment. That paradigm shift proves one thing, which is that structured automation is no longer an option.
A lab system transforms that dynamic in a different way. It is a dedicated platform that can control samples and manage data centrally and push laboratory workflows off of a controlled environment.
Essentially, a laboratory information management system serves as the electronic core of the operations. It also records samples, combines instruments and automatically keeps tamper-proof logs.
Key takeaway? The transition to the adoption of laboratory software by labs means the shift of the process of firefighting to the process of controlled, measured performance.
Top 5 Ways LIMS Software Optimizes Workflow
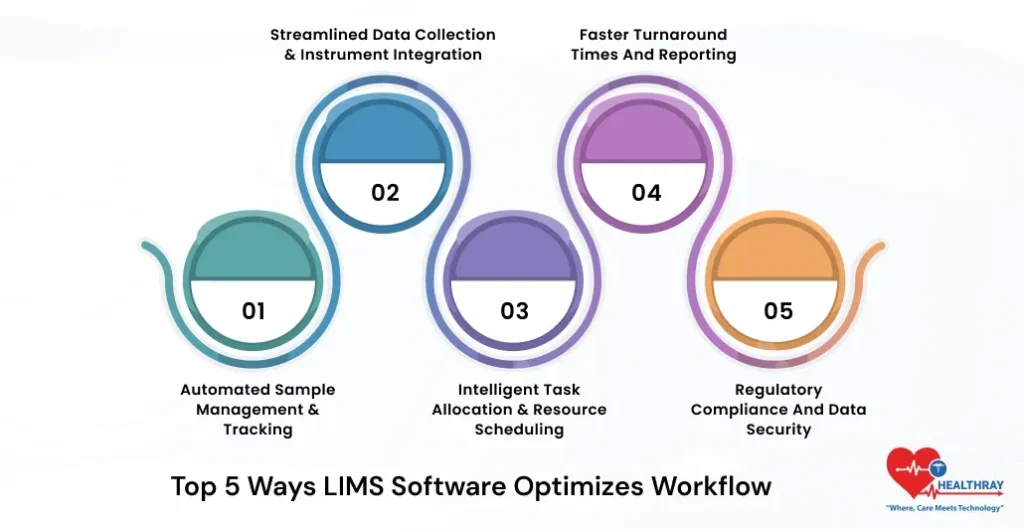
1. Automated Sample Management & Tracking
Operation control is based on sample visibility. Without structured tracking, mistakes multiply quickly.
A laboratory information management system assigns unique barcode identifiers at registration. From that moment, the LIMS system tracks who handled the sample, when testing occurred, and where it moved.
The chain of custody becomes transparent. Lost samples disappear. False labelling greatly decreases. Furthermore, a strong LIMS sample tracking system strengthens accountability across departments in both INDIA and the USA.
tamper-proof logs automatically save each action. As a result, there is an enhanced level of audit readiness.
2. Streamlined Data Collection & Instrument Integration
Manual transcription wastes time. It encourages errors, which are avoidable, even worse.
A laboratory software connects directly to instruments such as HPLC and mass spectrometers. Instead of copying values, LIMS software transfers results instantly.
The latter process safeguards data integrity and eliminates the risk of re-entry.
Moreover, the system compares the results with specifications and checks them at once. When results are out of tolerance, alerts are generated immediately on the teams.
Automated instrument integration can reduce transcription errors by more than 95% in structured environments.
Since compliance requirements in INDIA and the USA require traceability, this automation increases regulatory confidence.
3. Intelligent Task Allocation & Resource Scheduling
Inefficiency exists due to lack of coordination in tasks. Instruments sit idle. Reagents expire unused. Teams duplicate efforts.
A daily worklist is created with a laboratory system. Managers delegate tasks in a strategic and not in a reactive manner.
Concurrently, the calibration management schedules give warning when instruments are faulty. Emergency downtime is substituted by preventive maintenance.
Role-based access control is a restrictive feature that makes sure that unauthorized employees do not give consent to vital procedures. Thus, there is enhanced accountability between departments.
Such a systematic distribution minimizes workflow bottlenecks and enhances productivity in general.
4. Faster Turnaround Times and Reporting
Competitiveness is subject to speed. Customers want to get proper results immediately.
A laboratory information management system produces Certificates of Analysis immediately. Teams review results using electronic signatures instead of physical paperwork.
Upon approval, the platform disseminates reports safely amongst stakeholders. As a result, turnaround time shortens dramatically.
Automated reporting removes repetitive administrative processes as compared to manual compilation.
Research in the industry has shown that the laboratories that implemented cloud-based LIMS solutions realize 50 to 70 percent productivity gains.
5. Regulatory Compliance and Data Security
Non-compliance is not an option. Auditors demand orderliness and traceability.
A laboratory information management system maintains secure audit trails aligned with ISO 17025 and FDA electronic record regulations. Every modification records automatically. Every user action remains traceable.
Secure storage protects sensitive information. Structured permissions enforce role-based access control. Tamper-proof logs prevent unauthorized changes.
Whether operating in INDIA or the USA, laboratories must demonstrate documented control. The laboratory system provides that control at all times.
The Anatomy of an Optimized LIMS System Workflow
Let’s make this practical.
The vast majority of labs do not understand the level of losses they are making until they do a compare and contrast before and after.
1. Sample Registration
Prior to implementation, teams are making entries in paper logbooks or spreadsheets. Staff manually assign IDs. Errors happen quietly.
Barcodes are scanned immediately after the adoption of a laboratory information management system by technicians. The LIMS system uses automatically assigned identifiers. At that point, the chain of custody kicks off.
Traceability will therefore be automatic rather than reactive.
2. Testing Phase
In earlier times, instrument results were copied manually by the analysts. Such a practice was time consuming and compromised data integrity.
Lab software In lab software, instruments feed the results into LIMS software. The platform authenticates real-time information.
Inaccuracies are reduced to a minimum. This is because productivity is improved without employing a greater number of personnel.
3. Review & Validation
In conventional settings, supervisors put their signatures on paperwork. The paper flows across the departments. Delays follow.
Electronic signatures substitute the delay of modern laboratory software. Approvers confirms results in minutes of time.
As such, there is enhanced compliance and faster workflow.
4. Reporting
Formatting and verification in a manual compilation used to take hours.
The laboratory information management system is now capable of generating Certificates of Analysis automatically. The distribution occurs safely and immediately.
Highlights to Look for in a Modern Laboratory System
The adoption of technology is a matter of choice. Platforms do not offer the same value.
1. Cloud-Based Accessibility
Cloud-based lims platforms allow secure remote access in INDIA and the USA. Teams review data in real time without physical presence. As a result, collaboration improves across departments.
2. No-Code Configurable Workflows
Laboratories are constantly changing. A fixed system develops frustration. A powerful lab management system software package enables workflow setup without programmers. Processes are modified by managers within the company rather than depending on developers. Such flexibility does not allow stagnation of operations.
3. Instrument Integration Capabilities
Scalability is characterized by integration. There is a strong information management system in a laboratory that links with both old and new instruments. Seamless communication protects data integrity and eliminates re-entry errors.
4. Dashboards and Analytics Tools
Leaders need visibility. An advanced lab system will have dashboards that indicate the workflow bottlenecks and KPIs. Managers are active and not reactive.
Evidence-based supervision enhances efficiency and adherence.
Steps to Successful Laboratory Software Implementation
Technology is by itself not a guarantee of success. Strategy is a defining factor of result.
Step-1: Define Objectives
To begin with, pinpoint areas of operation pain. Determine whether your focus centers on turnaround time, compliance, or productivity. Effective configuration is based on clear objectives.
Step-2: Map Current Workflows
Document existing processes before making them digital. Know where the delays arise. Identify workflow bottlenecks honestly. Only then should the laboratory information management system replace manual steps.
Step-3: Choose the Right Vendor
Industry matters. The USA has various regulations compared to those of INDIA in pharma labs and food testing labs. Choose a vendor that has experience in the domain.
Step-4: Train Staff
Adoption determines ROI. Employ a developed train-the-trainer technique. Strengthen internal agents who facilitate wider implementation.
Step-5: Start with an MVP
Avoid overwhelming teams. Install the necessary modules. Grow slowly with increasing trust.
In many cases, labs that implement cloud-based LIMS solutions strategically report productivity improvements between 50% and 70%.
Healthray – A Smarter Approach to Laboratory Workflow Optimization
Now let’s talk about execution.
Healthray provides a full-fledged laboratory information management system created under the lab pressure. The platform is not imposed to have specific templates, but rather it is adapted to clinical, pharma, food, and environmental labs.
The system is connected with pathology lab software which is smooth to the system to maintain the labs with smooth reporting.
Moreover, the site integrates well with any contemporary laboratory computerized setting, and it can facilitate organized automation through registration to reporting.
Healthray has also advanced lab management system software capabilities that increase the data integrity, audit preparedness and clarity of operation.
Due to the importance of compliance, the system is in compliance with the regulations of FDA electronic records and ISO standards. All the transactions are secured by the role-based access control and tamper-proof logs.
Regardless of whether your lab is in INDIA or in the USA, Healthray enables digital transformation of scale.
Conclusion
Let’s be direct.
Competitive laboratories no longer have a luxury in the form of a laboratory system. Automation is a strategic requirement as a result of market growth, regulatory pressure and increased complexity of data.
Paperwork reduces productivity. Fragmented systems create workflow bottlenecks. Audit risk is increased by reactive operations.
On the other hand, a laboratory software concentrates its operations, enhances traceability, supports electronic signatures, and imposes role-based access control within the departments.
Consequently, there is an increment in efficiency, compliance preparedness as well as visualization in operations of laboratories in INDIA and USA.
In short, a LIMS system is an investment to bring about a structured expansion, enhance data integrity and future proofing of laboratory operations in a more highly regulated environment.
When you are interested in being able to have a structured workflow, as opposed to daily operational chaos, this is your time.
Book a free demo today and see how Healthray’s laboratory information management system transforms lab performance.